|
12/3/2023 0 Comments Entropy formula 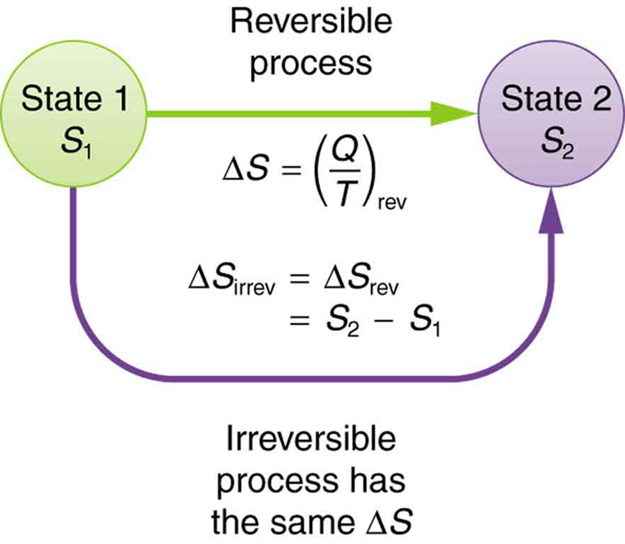
In 1934, Swiss physical chemist Werner Kuhn successfully derived a thermal equation of state for rubber molecules using Boltzmanns formula, which has since come to be known as the. (This equation in effect provides a thermodynamic definition of temperature that can be shown to be identical to the conventional thermometric one. In short, the Boltzmann formula shows the relationship between entropy and the number of ways the atoms or molecules of a thermodynamic system can be arranged. Although this result was obtained for a particular case, its validity can be shown to be far more general: There is no net change in the entropy of a system undergoing any complete reversible cyclic process. The test begins with the definition that if an amount of heat Q flows into a heat reservoir at constant temperature T, then its entropy S increases by S Q/T. Qualitatively, entropy is simply a measure how much the energy of atoms and molecules become more spread out in a process and can be defined in terms of statistical probabilities of a system or in terms of the other thermodynamic. There is no net change in the entropy of the Carnot engine over a complete cycle. Entropy is a state function that is often erroneously referred to as the 'state of disorder' of a system. To do so, the transmitter sends a series (possibly just one) partial messages that give clues towards the original message. It is represented by S, but in the standard state, it is represented by S°. The student will learn Entropy Formula with examples here. The concept of entropy basically talks about the spontaneous changes occurring in the everyday phenomenon or the tendency of the universe towards the disorder. 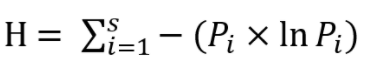
However, we know that for a Carnot engine, Entropy (Information Theory) In information theory, the major goal is for one person (a transmitter) to convey some message (over a channel) to another person (the receiver ). Thus, it can be applied in various stages or instances in a thermodynamic system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
